Stem cell therapy for cornea has emerged as a groundbreaking solution for individuals suffering from severe corneal injuries, which can often lead to vision loss and persistent discomfort. This innovative treatment utilizes cultivated autologous limbal epithelial cells (CALEC), which are harvested from a healthy eye and transplanted into the damaged cornea. In recent clinical trials, this therapy has shown impressive results, offering a beacon of hope for corneal repair where traditional methods fall short. With more than 90% efficacy in restoring the corneal surface observed over an 18-month period, patients are experiencing significant relief and improved vision restoration. As research continues to evolve, stem cell therapy for cornea could redefine eye injury treatment, providing previously unattainable options for those with chronic eye conditions.
Corneas that have suffered significant trauma or injury pose a considerable challenge in the realm of ophthalmologic care. The application of regenerative medicine techniques, specifically utilizing stem cell approaches, is paving the way for new treatments that could revolutionize eyesight rehabilitation. By employing limbal stem cells and advanced transplantation methods, researchers are actively developing experimental eye therapies designed to restore functionality to damaged corneal tissues. This innovative approach holds the promise of transforming the vision restoration landscape, offering a glimmer of hope for patients who have experienced limbal stem cell deficiencies. As the field of eye care continues to advance, the potential for these cutting-edge treatments to become mainstream could greatly alleviate the burden of visual impairments.
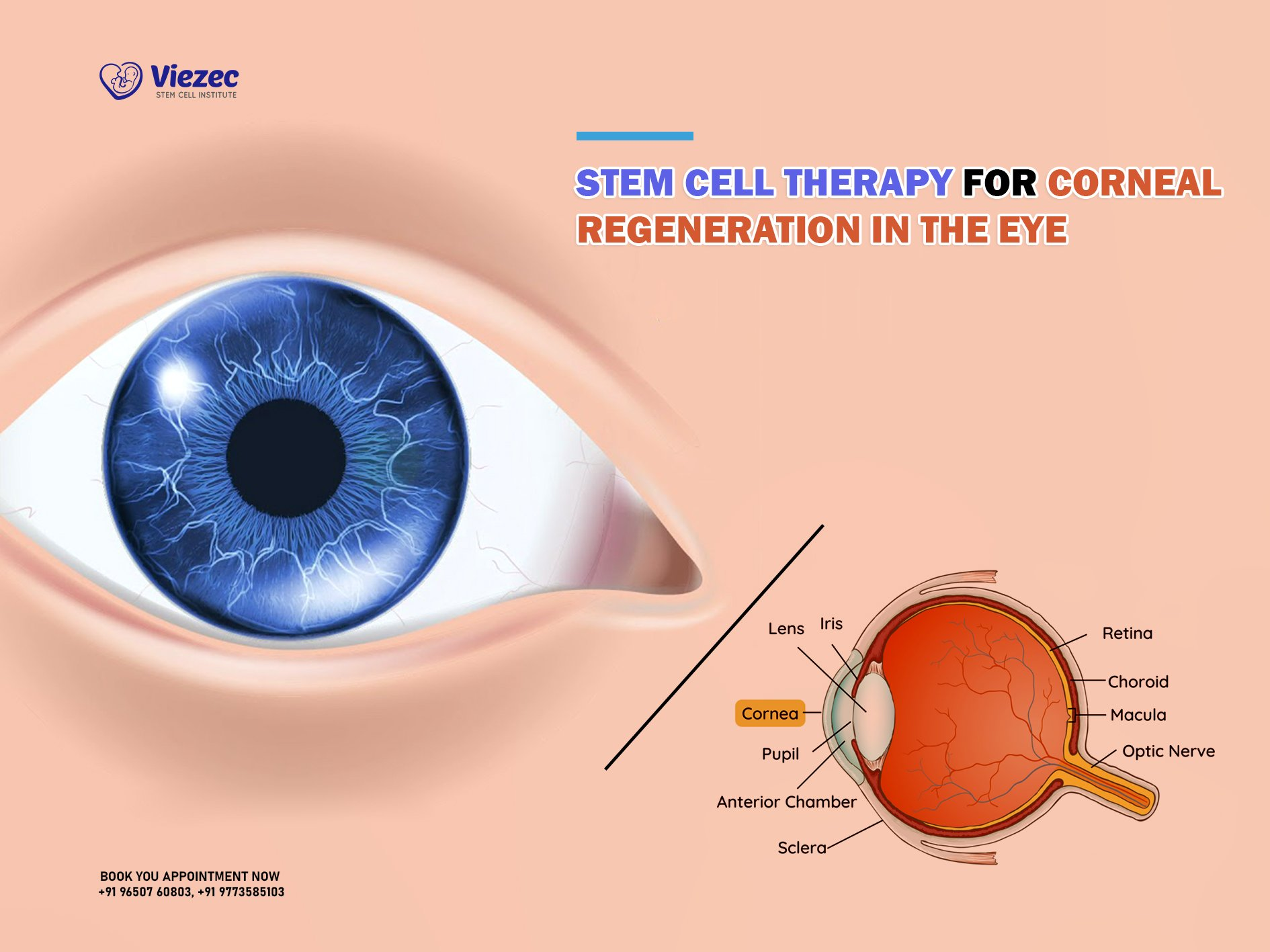
Understanding Limbal Stem Cells and Their Role in Corneal Repair
Limbal stem cells are critical components of the cornea, situated at the outer edge known as the limbus. They are essential for maintaining the health and integrity of the corneal surface, replacing damaged epithelial cells and providing a smooth barrier to protect the eye from environmental damage. When injuries occur, such as those from chemical burns or traumatic events, the depletion of these stem cells can lead to what’s known as limbal stem cell deficiency (LSCD), which hampers the eye’s ability to heal itself effectively. This deficiency can result in serious complications, including chronic pain and significant vision impairment.
The process of regenerating limbal epithelial cells provides hope for patients suffering from LSCD. Innovative therapies, such as stem cell therapy for cornea, aim to restore the function of these vital cells. Various techniques, including the cultivation of autologous limbal epithelial cells, have shown promise in clinical trials, demonstrating significant potential to repair the cornea and improve the quality of life for individuals previously deemed untreatable. By focusing on the regenerative capabilities of limbal stem cells, researchers explore new, effective pathways for corneal repair.
Innovative Stem Cell Therapy for Treating Eye Injuries
Stem cell therapy offers a groundbreaking approach to treating severe eye injuries that have traditionally left patients with little hope for recovery. The cultivated autologous limbal epithelial cell (CALEC) therapy introduced by Mass Eye and Ear is a prime example of this innovative treatment. During the CALEC procedure, a small sample of healthy limbal stem cells is harvested from the patient’s unaffected eye, cultivated in a specialized laboratory to expand into a graft, and then transplanted into the injured eye. This method not only restores the necessary stem cells but also promotes the regeneration of a functional corneal surface.
Clinical trials have shown that this experimental eye therapy yields promising results, with a significant percentage of participants experiencing substantial improvements in corneal health and visual acuity. The ability of CALEC to restore the cornea in long-term follow-ups emphasizes its potential as a viable treatment for conditions linked to corneal damage. The success of such therapies may drive further research and development, paving the way for widespread adoption in clinical practice and offering renewed hope for vision restoration in patients previously considered at risk of permanent damage.
Although still an experimental procedure, the encouraging outcomes from initial clinical trials have sparked interest in expanding the reach of stem cell therapies for eye injuries. Future studies aim to include larger patient cohorts and assess the efficacy of similar approaches in treating both eyes, using stem cells from cadaveric donors. Such developments could revolutionize the standard of care in treating corneal injuries, making vision restoration a reality for more individuals suffering from debilitating eye conditions.
The Future of Corneal Restoration Through Stem Cell Research
As research advances, the horizon looks promising for expanding treatment options in corneal repair, particularly through the cultivation of limbal stem cells. The initial findings from the CALEC clinical trials, showcasing a 90 percent success rate in restoring the corneal surface, open new avenues for investigation and potential therapeutic applications. The National Eye Institute’s funding supports not only the immediate goals of the CALEC project but also encourages innovative thinking about how limbal stem cells can be leveraged to treat a variety of corneal ailments.
Future studies will likely focus on scaling the manufacturing process for CALEC grafts and exploring allogeneic sources of limbal stem cells. This could effectively expand treatment eligibility to patients with bilateral corneal damage. By addressing these challenges, researchers aim to transform the landscape of eye injury treatment, establishing a solid foundation for widespread clinical implementation of this groundbreaking technology. With ongoing investigations delivering positive results, the objective remains clear: to facilitate access to effective, life-changing therapies for individuals struggling with severe corneal injuries.
Clinical Trial Success and Patient Outcomes in Corneal Treatments
The clinical trials conducted for CALEC therapy have not only revealed significant success rates but also demonstrated a high safety profile for patients involved. As observed, a remarkable 93 percent success rate at the twelve-month mark is a testament to the efficacy of using cultivated limbal stem cells for repairing corneal ailments. These outcomes highlight how innovative stem cell treatments can shift the paradigm of care for individuals facing debilitating eye conditions, providing them with a renewed quality of life and restored vision.
Patient feedback from these trials reflects the profound impact of this therapy on their everyday lives. Many participants report drastic improvements in visual acuity, allowing them to partake in activities that were previously hindered by their corneal damage. Such outcomes not only validate the scientific approach behind stem cell therapy for cornea but also reinforce the importance of continued research and trials. As more compelling data emerges, it emphasizes the urgent need for broader acceptance and integration of these regenerative therapies into standard ophthalmic care.
Challenges in Implementing Stem Cell Therapy for Cornea
Despite the promising results associated with stem cell therapy for cornea, several challenges remain in the implementation of this innovative treatment. One of the primary concerns is the need for a single unaffected eye to procure limbal stem cells, which limits patient eligibility for the therapy. Additionally, the processes involved in graft manufacture and subsequently transplanting them into the damaged eye require stringent quality control and regulatory approvals, which can delay wider application even when initial studies show promise.
Researchers are now considering the development of allogeneic sources for limbal stem cells, which could potentially address the limitations of current practices. By sourcing stem cells from cadaveric donors, the goal is to create a viable supply that can cater to patients suffering from bilateral eye involvement. While this avenue presents its own set of ethical and regulatory hurdles, overcoming these challenges will enhance the reach of stem cell therapy and ideally lead to an effective, standard treatment option for more patients facing severe corneal injuries.
The Role of Regulatory Bodies in Advancing Eye Therapy Research
The progression of stem cell therapy for eye injuries is significantly influenced by the role of regulatory bodies such as the U.S. Food and Drug Administration (FDA). The approval process for experimental therapies is crucial as it ensures that treatments are both safe and effective before becoming widely available. The CALEC clinical trials exemplify the rigorous scrutiny that these innovative treatments must undergo to gain public trust and acceptance. Enhanced collaboration between research institutions and regulatory agencies is essential to streamline this process and facilitate timely interventions for patients in need.
Research funding, particularly from entities like the National Eye Institute, plays an indispensable role in propelling advancements in experimental eye therapy. By providing necessary resources and backing for clinical trials, these organizations enable researchers to explore groundbreaking methodologies, leading to potential new therapies that could transform current treatment paradigms. Continuous engagement from regulatory bodies and funding agencies will be vital in ushering in new approaches to corneal repair, ultimately benefiting patients with eye injuries.
Patient Perspectives on Innovations in Eye Injury Treatments
The experiences and perspectives of patients undergoing innovative treatments such as stem cell therapy for cornea are invaluable in shaping future research and development. Many participants in the CALEC clinical trials have expressed hope and optimism regarding their conditions, citing improvements not just in vision but also in overall quality of life. The narratives from these patients serve to underscore the significance of such treatments, illustrating the tangible benefits that can emerge from cutting-edge medical interventions.
Moreover, patient involvement in the research process brings about a unique viewpoint, emphasizing the emotional and psychological dimensions of dealing with severe eye injuries. Understanding these dimensions can guide researchers and medical professionals in refining approaches to care, ensuring that therapies not only restore vision but also support the holistic well-being of individuals. As stem cell therapy continues to evolve, including patient voices in the dialogue will be crucial to driving meaningful change within the field of ophthalmology.
The Importance of Collaborative Research in Eye Care
Collaborative research efforts, as seen with the involvement of multiple institutions like Mass Eye and Ear, Dana-Farber, and Boston Children’s Hospital, are paramount in advancing treatments for corneal injuries. This interdisciplinary approach fosters innovation, allowing researchers to pool their expertise and resources to tackle complex challenges associated with stem cell therapy for cornea. Such collaborations not only enhance the design and implementation of clinical trials but also promote a more comprehensive understanding of treatment outcomes and their implications for patient care.
Moreover, the synergy created by working across different healthcare sectors accelerates the transition of promising therapies from the lab to real-world applications. By collaborating on shared goals, these institutions can drive significant advancements in eye care, ultimately shortening the timeline for potential treatments to reach the market. The ongoing commitment to collaborative research is crucial for unlocking the full potential of stem cell therapies and ensuring that innovative solutions are available to patients in desperate need of effective eye injury treatments.
Future Directions for Limbal Stem Cell Therapy
Looking ahead, the potential of limbal stem cell therapy is immense, with research set to explore various avenues aimed at enhancing the efficacy and accessibility of these treatments. Ongoing studies will focus on optimizing the manufacturing process of CALEC grafts, with the aspiration of producing larger quantities suitable for broader patient demographics. Innovations in stem cell cultivation techniques and improved methodologies for transplant procedures may also contribute to enhanced success rates and patient outcomes.
Additionally, researchers are likely to delve into the genetic factors and biological mechanisms that govern limbal stem cell behavior, seeking to unlock further advancements in corneal repair. As knowledge in regenerative medicine expands, the potential for developing new therapies that capitalize on these insights could revolutionize the treatment of corneal injuries once thought unmanageable. The future of stem cell therapy for cornea holds considerable promise, and continued investment in this frontier of eye care will be essential to translating research advances into clinical success.
Frequently Asked Questions
What is stem cell therapy for cornea and how does it work?
Stem cell therapy for cornea involves using cultivated autologous limbal epithelial cells (CALEC) to restore damaged corneal surfaces. A biopsy from a healthy eye retrieves limbal stem cells, which are then expanded into a graft and transplanted into the affected eye. This process can effectively treat severe corneal injuries, enhancing the potential for vision restoration.
What conditions can stem cell therapy for cornea address?
Stem cell therapy for cornea can address conditions such as corneal injuries resulting from chemical burns, infections, or trauma that lead to limbal stem cell deficiency. This therapy offers new hope for patients with irreversible corneal damage previously deemed untreatable.
What is the success rate of stem cell therapy for cornea as per recent trials?
Recent clinical trials of stem cell therapy for cornea have shown impressive results, with more than 90 percent effectiveness in restoring the cornea’s surface. At the 18-month follow-up, complete corneal restoration was achieved in 77 percent of participants, indicating significant progress in corneal repair.
Is stem cell therapy for cornea considered safe?
Yes, stem cell therapy for cornea has demonstrated a high safety profile in clinical trials, with no serious adverse events reported. One minor issue related to a bacterial infection was documented, but it was resolved quickly, reinforcing the potential safety of this experimental eye therapy.
Can stem cell therapy for cornea be offered to patients with both eyes injured?
Currently, stem cell therapy for cornea can only be performed on patients with one involved eye, as a healthy eye is used for the required biopsy. Future research aims to develop an allogeneic manufacturing process using limbal stem cells from cadaveric donor eyes, potentially widening access to those with bilateral eye damage.
How long does it take to see results from stem cell therapy for cornea?
Patients undergoing stem cell therapy for cornea may start noticing improvements in corneal healing within three months, with continued improvement observed over time. Follow-up visits at 12 and 18 months showed sustained success rates in corneal surface restoration and visual acuity enhancement.
What is the next step for stem cell therapy for cornea after clinical trials?
Following promising clinical trial results, further studies are necessary to expand patient numbers, ensure longer follow-ups, and refine treatment protocols. Once sufficient data is gathered, researchers aim to seek FDA approval for broadening access to stem cell therapy for cornea.
What role do limbal stem cells play in corneal health?
Limbal stem cells, located at the limbus of the cornea, are crucial for maintaining the eye’s surface health. They regenerate epithelial cells that protect the cornea and ensure proper vision. A deficiency in these stem cells, often due to injury, can lead to severe vision impairment.
Who conducted the clinical trials for stem cell therapy for cornea?
The clinical trials for stem cell therapy for cornea were conducted by Mass Eye and Ear, led by Ula Jurkunas, in collaboration with researchers from Dana-Farber and Boston Children’s Hospital. This study marked a significant advancement in experimental eye therapy.
Why is stem cell therapy for cornea considered experimental?
Stem cell therapy for cornea is classified as experimental because it is still undergoing clinical trials to evaluate its long-term effectiveness and safety. Further studies are needed before this innovative treatment can be officially approved for widespread clinical use.
| Key Point | Details |
|---|---|
| Introduction of CALEC Surgery | Ula Jurkunas performs the first CALEC surgery at Mass Eye and Ear. |
| Stem Cell Therapy | Developed procedure utilizes stem cells from a healthy eye to restore a damaged corneal surface. |
| Clinical Trial Success | Trial showed over 90% effectiveness with significant improvement in corneal surface restoration. |
| Procedure Overview | Involves biopsy of healthy eye, developing CALEC graft over 2-3 weeks, and transplantation into damaged eye. |
| Safety Profile | No serious adverse events; only minor issues reported. |
| Future Developments | Aim to create allogeneic grafts for patients with damage to both eyes. |
| FDA Status | Considered experimental; further studies needed for federal approval. |
Summary
Stem cell therapy for cornea offers new hope for patients with blinding cornea injuries. The innovative CALEC procedure, developed at Mass Eye and Ear, successfully restores the cornea’s surface by using stem cells harvested from a healthy eye. In a recent clinical trial, 14 patients experienced a remarkable improvement in their condition, with over 90% achieving corneal restoration. This technique not only represents a significant advancement in treating previously untreatable eye injuries but also highlights the potential of stem cell therapies in ophthalmology. Further research is needed to expand this promising treatment, making it accessible to a larger population.